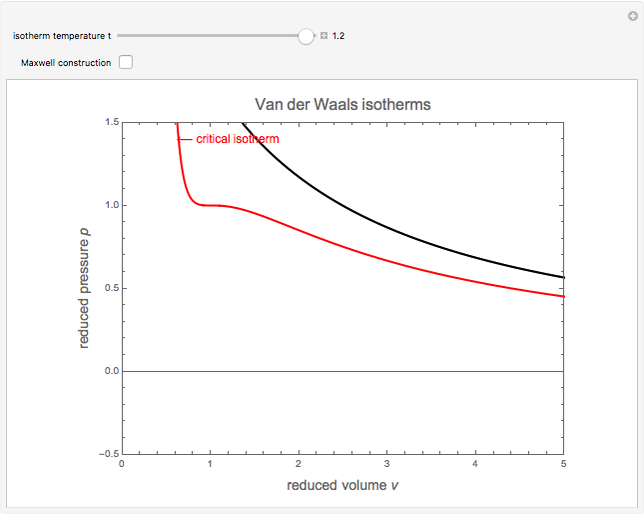
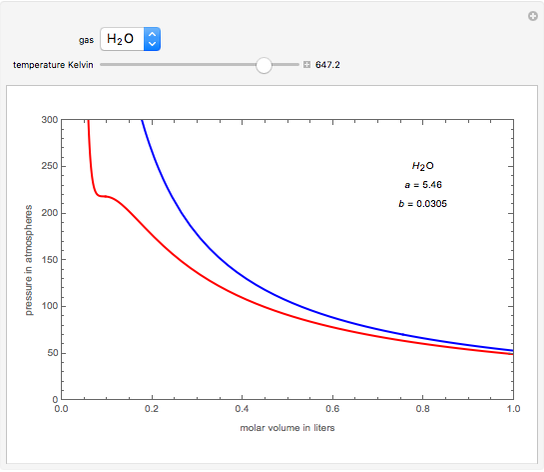
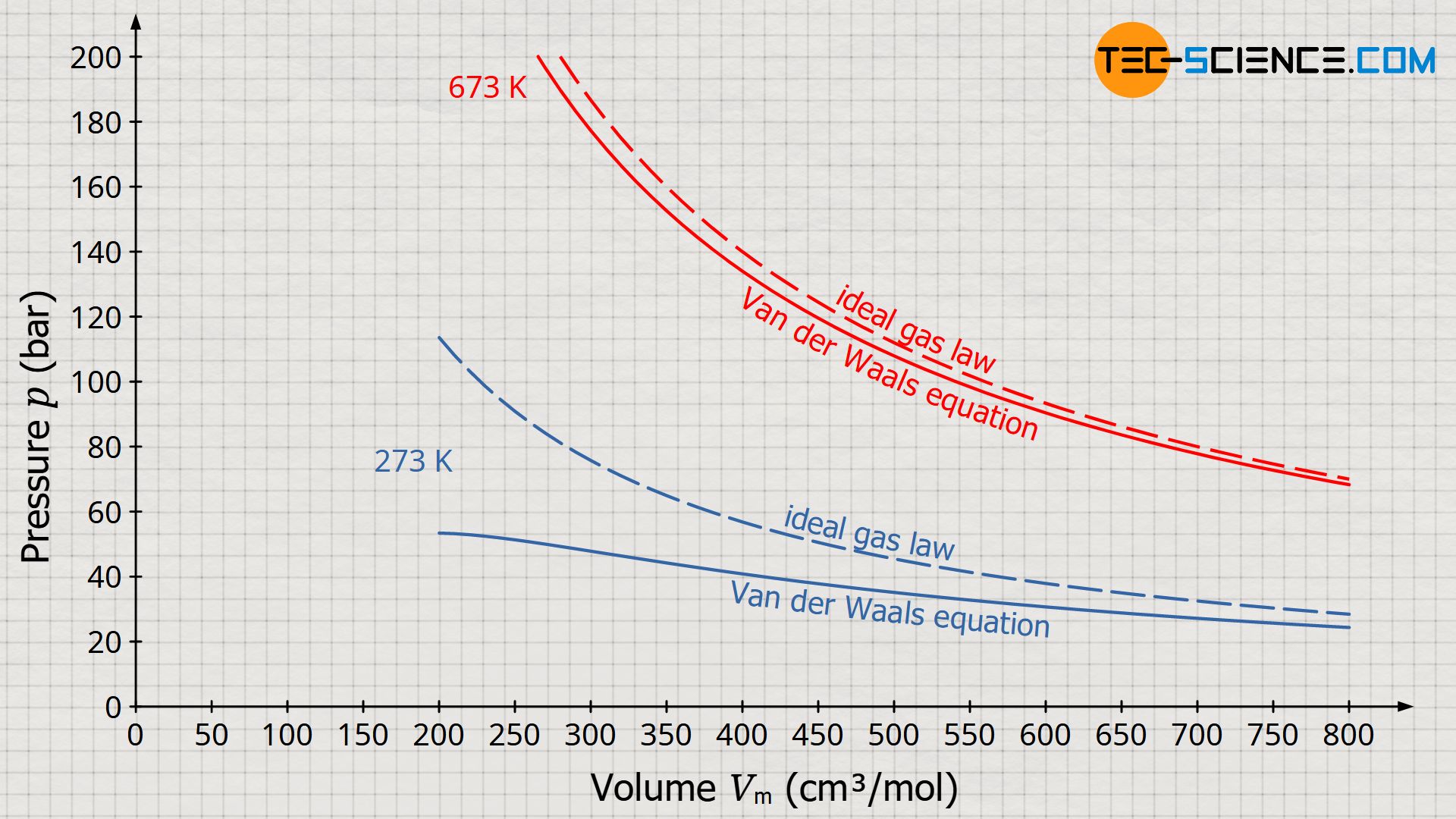
SOLVED: Equations van der Waals EoS RT P = VSEbIWi Redlich-Kwong EoS RT P = VnEb ViVn(n+ b) 17 dv B =vaZ)p 1v K = Ev ae)r dP av Cp = Gv+TSzlvGz)e
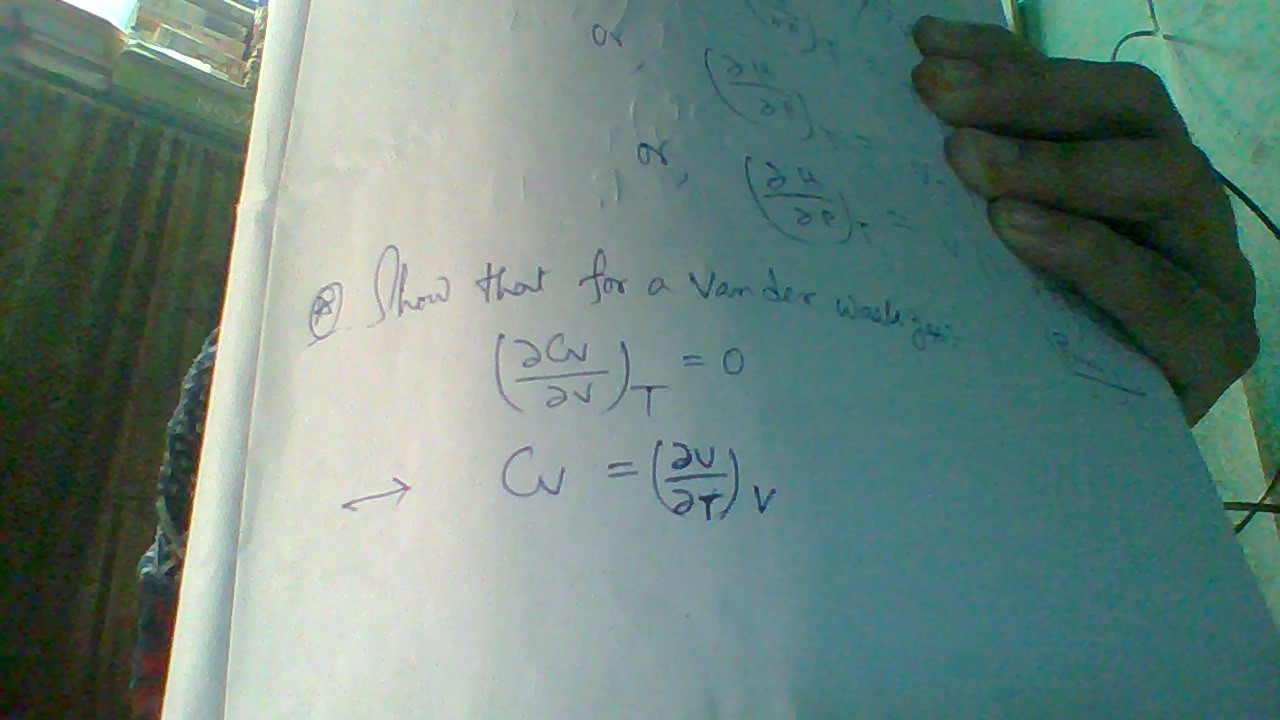
Show that for a van der Waals gas, ((delC_V)/(delV))_T = 0, where C_V = ((delU)/(delT))_V? | Socratic

The virial form of van der Waal's gas equation is PV = RT (1 + BV + CV^2 + .... ) = RT(1 + B'P + C'P^2 + ....) . The second

Find the entropy increment of one mole of a Van der Waals gas due to the isothermal variation of volume from V1 to V2. The Van der Walls corrections are assumed to

From van der Waals to VTPR: The systematic improvement of the van der Waals equation of state - ScienceDirect
Write the Van der Waals equation via the reduced parameters π, v, and τ, having taken the corresponding - Sarthaks eConnect | Largest Online Education Community