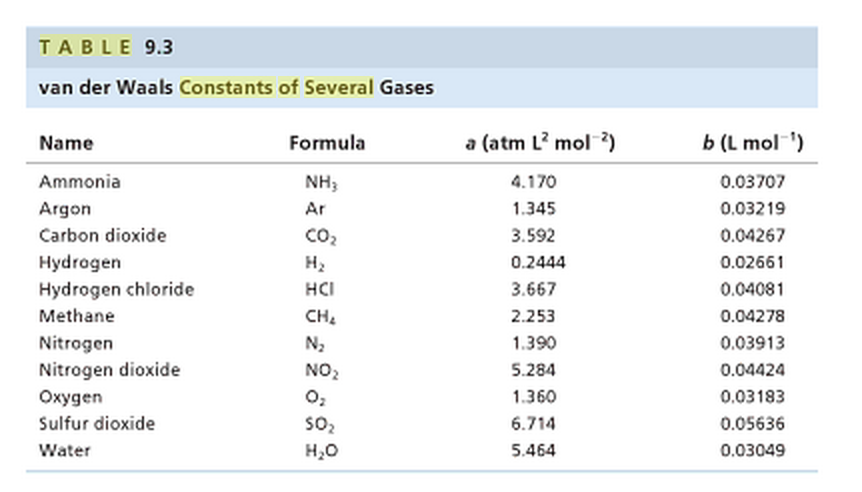
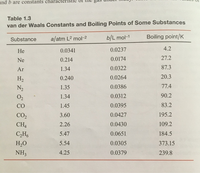
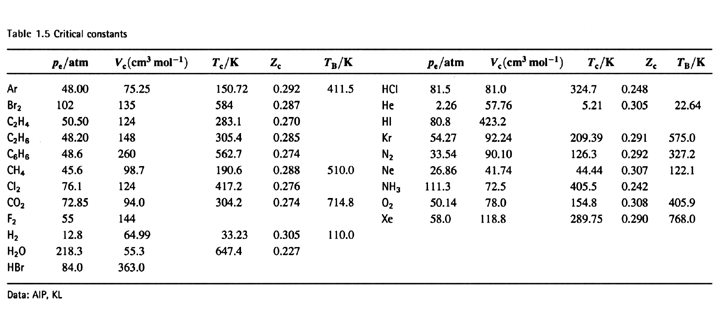
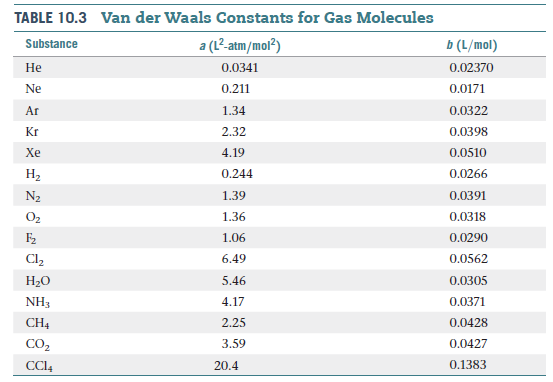
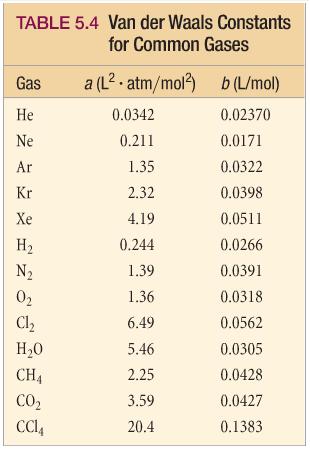
The van der Waals constant for gases A, B, and C are as follows Answer the following: Which gas has the most ideal behaviour around STP?

Find out the unit and dimensions of the constants `a and b` in the van der Waal\'s equation `( P... - YouTube

homework and exercises - Van der Waals constant $b$ (real gas) chemical form. only - Physics Stack Exchange
Derive the values of critical constants from the Van der Waals constants. - Sarthaks eConnect | Largest Online Education Community
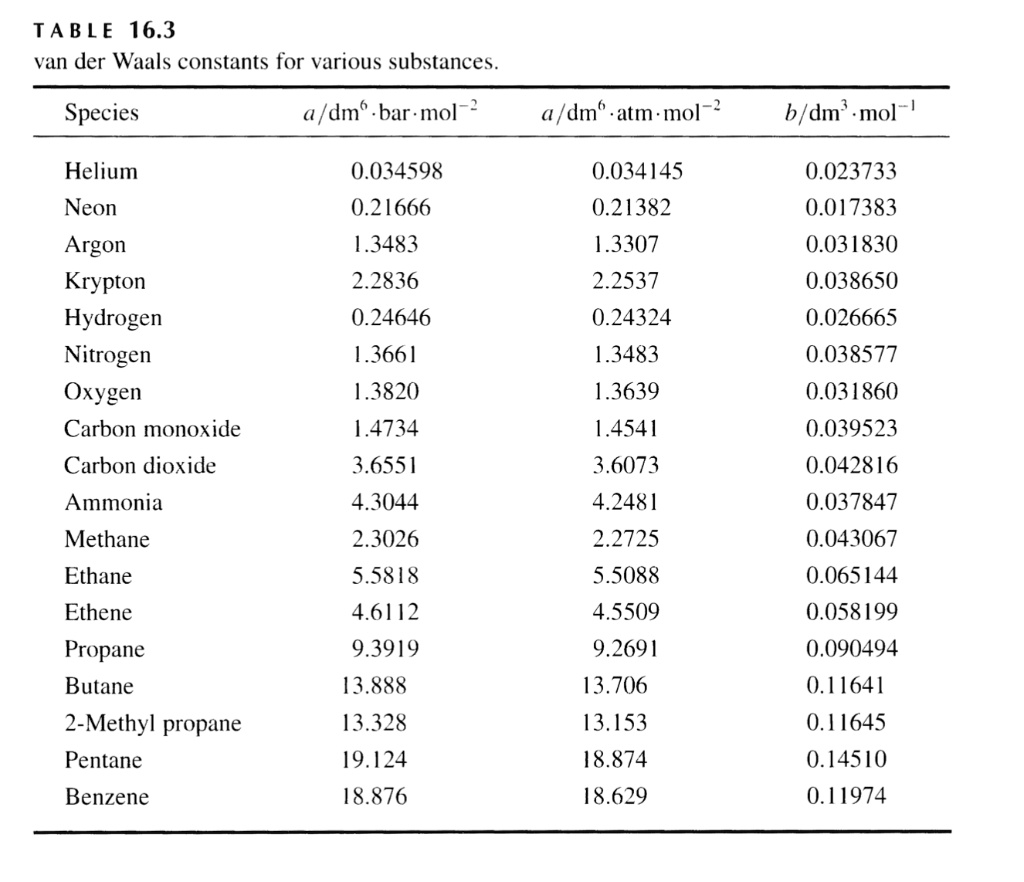
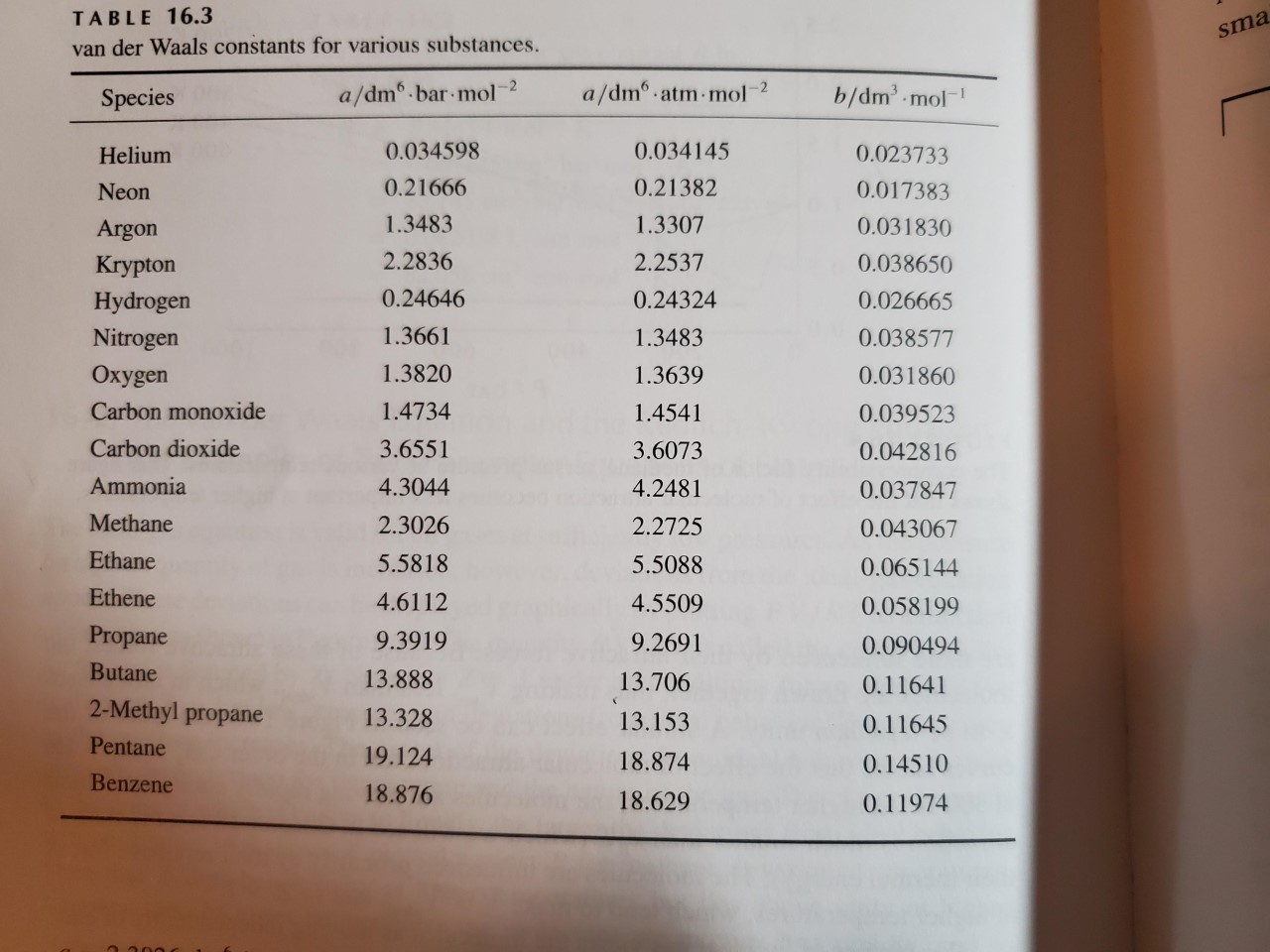
SOLVED: TA BL E 16.3 van der Waals constants for various substances Species /dm6 . bar-mol dm" . atm- mol b/dm'. mol Helium Neon Argon Krypton Hydrogen Nitrogen Oxygen Carbon monoxide Carbon